Is time on our side?: Micro-Entry
by Dr. Allan Bacon
Metabolically healthy obese status (sometimes termed the obesity paradox) is likely a transient condition that requires weight management to prevent the development of unhealthy cardiometabolic features in the future. Weight loss, but not gains in cardiorespiratory fitness after exercise-training, predicts improved health risk factors.Mora-Rodriguez et al. 2018
Although fitness is certainly important (and can improve/temper health risk factors to an extent), the number one indicator of improved cardiometabolic risk factors is weight loss. Believing that seemingly normal cardiometabolic blood levels alone are a case for “fit” is a misguided, and frankly dangerous, viewpoint in itself.
Research has shown that, more often than not, it is only a matter of time before degradation of joints, increased cancer prevalence, cardiovascular disease onset, loss of blood glucose control, and the formation of many other chronic health conditions develop if excess body fat is not lost.
In the research by Mora-Rodriguez et al. referenced above:
•MetS health improvements were associated with body weight losses rather than with cardio-respiratory fitness increases.
•Clinical advice should emphasize body weight loss in exercise programs.
One hundred sixty middle aged individuals with metabolic syndrome, with body mass index (BMI) of 32±5 kg m−2 completed this study. Metabolic syndrome (MetS) was defined as the presence of three of the following five risk factors: elevated waist circumference, blood pressure, fasting blood glucose, blood triglycerides, or reduced HDL-C. Exclusion criteria included untreated cardiovascular or renal disease, or any condition associated with exercise intolerance.
Numerous studies reinforce the fact that obesity, regardless of blood markers at a single point in time, is an extreme health risk. Gaiţă and Moşteoru (2017) put it best when they said: “Obesity presents one of the biggest issues of modern-day life for a wide variety of medical specialties, starting with the cardiologist and ending with the diabetologist or the bariatric surgeon.
However, in the last few years a new entity has emerged, the ‘metabolically healthy obese (MHO) individual’. This category encompasses obese individuals without the presence of metabolic diseases such as type 2 diabetes mellitus, dyslipidemia, or hypertension. Several studies have thus been undertaken to determine the risk of these MHO individuals for diabetes mellitus or cardiovascular diseases compared with the ‘unhealthy’ obese…one trend seems to be emerging: MHO is but a transient phase in the path toward insulin resistance and metabolic syndrome.”
Further Research
A separate study using resistance training bolstered the findings of the title study by showing fat loss was more effective for cardiometabolic health than muscle gain. “Reductions in fat mass predicted increases in HDL cholesterol, insulin sensitivity index, and decreases in waist circumference. In contrast, increases in lean mass did not predict changes in any of the measured cardiometabolic health indices.” The findings continued: “Health improvements with training that emphasize resistance exercises are typically attributed to increases in lean mass; however, these results underscore reducing body fat to predict cardiometabolic health improvements.”
https://www.sciencedirect.com/science/article/pii/S0271531718307735
“Body mass index (BMI), waist circumference (WC), percentage of fat mass (BF%), fat-mass index (FMI), and waist-to-height ratio (WHR) were included as body fatness measurements. A high cardio-metabolic risk cluster was derived by assessing triglycerides, low-density lipoprotein (LDL) cholesterol, high-density lipoprotein (HDL) cholesterol, fasting glucose, and blood pressure. In both sexes, higher levels of all fatness parameters were associated with increased cardio-metabolic risk. Combined analysis showed that unfitness (lower normalized grip strength) and high fat had the highest cardio-metabolic risk odds ratio for waist circumference and fat-mass index in men and women, respectively.”
https://www.mdpi.com/2072-6643/10/11/1742
Metabolically healthy obese status is likely a transient condition where weight management is demanded to prevent the establishment of unhealthy cardiometabolic features. Over half of the participants in this study (ATTICA study: 1514 men and 1528 women (aged >18 years old) free of CVD and residing in the greater Athens area, Greece) who were deemed “metabolically healthy obese” transitioned to cardiovascular disease or metabolic disorder onset within 10 years.
https://www.sciencedirect.com/science/article/pii/S0026049519300101
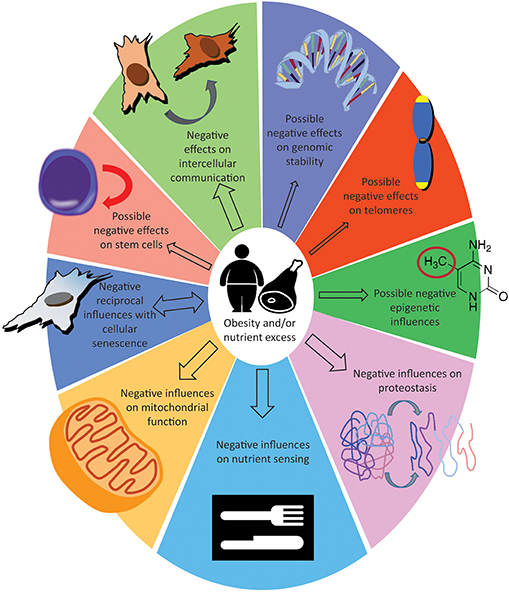
The literature persuasively suggests that the accumulation of pro-inflammatory cells, in the adipose tissue of obese patients, through cytokines and extracellular vesicles, accelerates the rate of aging both in the adipose tissue itself and the entire organism. Mitochondrial dysfunction occurs in aged tissues, in response to excessive nutrient intake, and in obesity, contributing to inflammation and insulin resistance. Aging and obesity appear superimposable in their impact on mitochondria and it is reasonable to hypothesize that they could exert additive effects. Obesity significantly decreases mechanisms associated with proteome maintenance. There appears to be a strong relationship between obesity and senescence. There is solid evidence that obesity deregulates cellular mechanisms related to nutrient sensing.
https://www.frontiersin.org/articles/10.3389/fendo.2019.00266/full
“Metabolically healthy obese individuals are associated with an increased risk of cardiovascular diseases.”
https://www.sciencedirect.com/science/article/abs/pii/S0167494319300111
About 35% of all subjects with obesity are “metabolically healthy”. These individuals have approximately half the risk of developing type 2 diabetes and cardiovascular disease compared with metabolically unhealthy subjects with obesity, but they still have a significantly greater risk (by 50–300%) compared with metabolically healthy lean subjects. Therefore, absence of metabolic risk factors in people with obesity should not be a contraindication for weight-loss treatment. Metabolically healthy obesity needs to be treated, and this need is reinforced by the fact that this phenotype is not stable over time, as ∼50% of these subjects will cease being metabolically healthy within ∼10 y. Intervening early is therefore important.
https://academic.oup.com/ajcn/advance-article-abstract/doi/10.1093/ajcn/nqz133/5523188
“Obesity is a convincing risk factor for 11 types of cancer.”
https://www.sciencedirect.com/science/article/pii/S0026049518302312
People who have obesity but no metabolic complications are likely to develop those complications later.
https://www.nature.com/articles/ijo2017233
Over time, people with overweight and obesity are more likely to develop cardiometabolic risk factors, with rising blood glucose being the most likely negative health outcome.
https://www.nature.com/articles/ijo2017249
“these flawed concepts of harmless obesity tend to support a bias for neglecting treatment.”
https://www.nature.com/articles/ijo2017311
“The detrimental effects of overweight/obesity may extend beyond the duration of overweight/obesity itself.”
https://onlinelibrary.wiley.com/doi/10.1002/oby.22819
“Almost one-third of the obese individuals are in metabolic health. However, they are still at higher risk of advancing to unhealthy state. Therefore, it is still needed to advise MHO individuals to maintain or adopt a healthy lifestyle, so as to counterbalance the adverse effects of obesity.”
DOI: 10.1097/MD.0000000000008838
“In conclusion, the present study reports a positive association between MHO and cancer incidence. All individuals with obesity, even in the absence of metabolic dysfunction, should be encouraged to lose weight.”
https://onlinelibrary.wiley.com/doi/10.1111/obr.13049
“This review aims to show how obesity and ageing are “two sides of the same coin” through discussing how obesity predisposes an individual to age-related conditions, illness, and disease. We will further demonstrate how the mechanisms that perpetuate the early-onset of chronic diseases in obesity parallel those of ageing.”
PMID: 32020741
“Metabolically healthy obesity is not a stable or reliable indicator of future risk for CVD. Weight loss and lifestyle management for CVD risk factors should be recommended to all individuals with obesity.”
PMID: 29699611
“Compared with metabolically healthy normal-weight individuals, obese persons are at increased risk for adverse long-term outcomes even in the absence of metabolic abnormalities, suggesting that there is no healthy pattern of increased weight.”
PMID: 24297192
“Weight management should be recommended to all people with obesity, irrespective of their metabolic status, to lower risk of diabetes, ASCVD, HF and respiratory diseases. The term ‘MHO’ should be avoided as it is misleading and different strategies for risk stratification should be explored.”
doi.org/10.1007/s00125-021-05484-6
“We agree with the perspective of Gaesser and Blair that an emphasis to optimize health should be placed on sustained engagement in lifestyle behaviors, which includes physical activity, in adults with obesity. However, we disagree that the emphasis should ignore obesity as an outcome and focus primarily on cardiorespiratory fitness. Rather, we suggest that to optimize health, the public health and clinical focus should be on lifestyle behaviors that influence both weight status and cardiorespiratory fitness.”
DOI: 10.1249/MSS.0000000000001747
“The long-term trajectories analysis showed that moderate PA may not decrease the risk of CVD in persistently overweight and obesity adults.”
PMID: 35090510
“In a large cohort study of metabolically healthy but overweight or obese adult men and women, weight reduction was associated with a lower incidence of subclinical carotid atherosclerosis. Our findings suggest that metabolically healthy overweight or obese subjects may benefit from weight reduction in terms of cardiovascular disease risk.”
https://www.ncbi.nlm.nih.gov/pubmed/31545359
“Obesity presents one of the biggest issues of modern-day life for a wide variety of medical specialties, starting with the cardiologist and ending with the diabetologist or the bariatric surgeon. However, in the last few years a new entity has emerged, the metabolically healthy obese (MHO) individual. This category encompasses obese individuals without the presence of metabolic diseases such as type 2 diabetes mellitus, dyslipidemia, or hypertension. Several studies have thus been undertaken to determine the risk of these MHO individuals for diabetes mellitus or cardiovascular diseases compared with the ‘unhealthy’ obese…one trend seems to be emerging: MHO is but a transient phase in the path toward insulin resistance and metabolic syndrome.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6768534/
“In MetS individuals, the exercise-training increases in cardiorespiratory fitness (CRF; estimated by VO 2max) are not predictive of the improvements in their health risk factors. Instead, body weight loss (<2%) was a significant contributor to the improved MetS Z-score and thus should be emphasized in exercise training programs."
doi:https://doi.org/10.1016/j.numecd.2018.08.004